A newly identified enzyme may help explain why patients with cancer experience severe muscle loss—and could point to a new therapeutic strategy.
Muscle wasting, a hallmark of cancer cachexia, is a common and debilitating condition affecting up to half of patients with advanced cancer. It is associated with weakness, fatigue, reduced treatment tolerance, and poorer survival, yet no approved therapies currently exist.
In a study published April 25 in the Journal of Clinical Investigation, researchers from Duke University School of Medicine and Duke-NUS Medical School, Singapore, discovered that an enzyme called MESH1 plays a central role in driving this process.
MESH1 levels were found to be elevated in muscle samples from patients with cancer-associated wasting. Reducing the activity of MESH1 in muscle cells protected the cells from ferroptosis, a form of oxidative cell death.
“This gives us a tangible path to intervene and that’s something we didn’t have before,” said Jen-Tsan Chi, PhD, a cancer biologist and professor in the Duke Department of Molecular Genetics and Microbiology.
The team identified MESH1 as the long-sought enzyme that breaks down coenzyme A (CoA), a molecule essential for energy production and cellular stress protection. Muscles samples from patients with cancer-related muscle loss had elevated levels of the enzyme.
“Naming MESH1 as that enzyme turns CoA degradation into a regulated, druggable lever and our data show that lever is turned up in the muscle of those with cancer-related muscle loss,” said Chi, co-leader of the Cancer Biology Program at Duke Cancer Institute.
In experiments using fruit flies, excess MESH1 levels reduced the number of helpful molecules that protect cells, triggering muscle degeneration. But when the team blocked the effect of MESH1—either through genetic modifications or administering drugs—the muscles stayed healthier, and, in some cases, reversed wasting.
“This pathway helps explain why muscle becomes fragile in patients with cancer cachexia,” said co-corresponding author Hong-Wen Tang, PhD, an assistant professor in the Cancer & Stem Cell Biology Signature Research Programme at Duke-NUS Medical School, Singapore. “Understanding changes in components of the pathway could be key to restoring muscle strength and physical function.”
The Duke-NUS team conducted preclinical studies and analyses of human tissue samples to investigate how MESH1 impacts muscle function in patients with cancer cachexia.
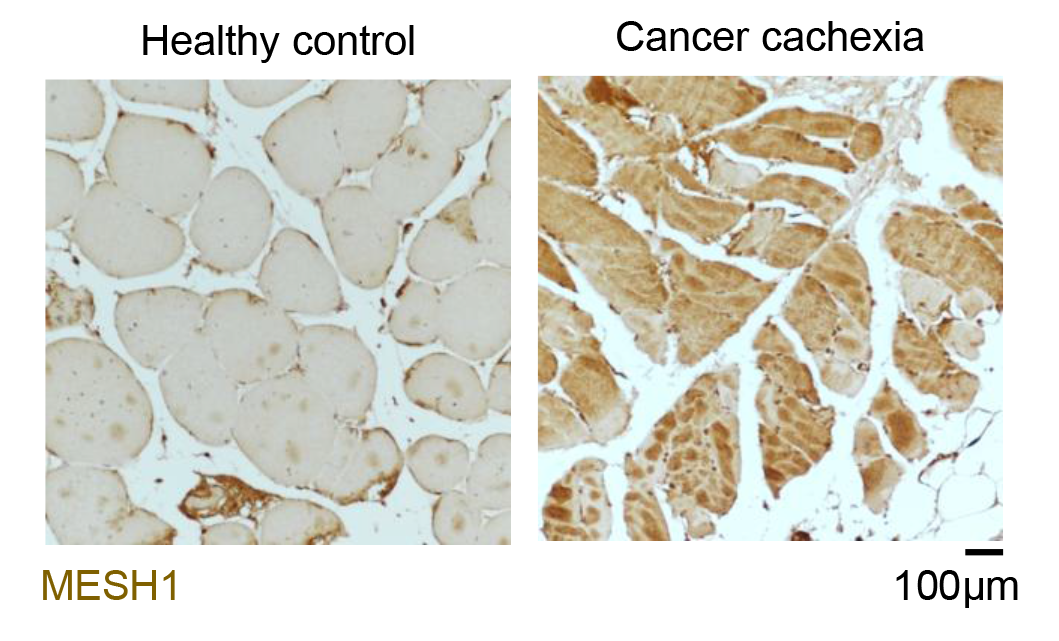
Human Muscle tissue samples were stained brown to show where the MESH1 enzyme is present—the darker the brown, the more MESH1. The left image shows muscle from a healthy person without muscle wasting, with light brown staining and healthy, round muscle cells. The right image shows muscle from a cancer patient with muscle wasting, with much darker brown staining and visibly shrunken, damaged muscle cells, suggesting that too much MESH1 may be actively contributing to muscle loss in cancer patients. // Image credit: Goh Kah Yong, Duke-NUS Medical School
An enzyme with a double life
MESH1 wasn’t completely unknown before the study. Scientists already knew it breaks down another important molecule, NADPH, which helps protect cells from stress and damage.
What surprised researchers is that MESH1 does both jobs — breaking down NADPH and CoA — giving it powerful control over whether cells survive or die.
“How MESH1 acts as a master regulator of multiple metabolic pathways remains mysterious,” said Pei Zhou, PhD, professor of biochemistry at Duke and co-corresponding author. “AI-based protein engineering may ultimately enable the design of variants with isolated functions, helping us dissect its individual roles.”
The findings point to MESH1 as a promising drug target. In theory, inhibiting the enzyme could help maintain CoA levels and shield muscle tissue, even as patients receive aggressive therapies such as chemotherapy and radiation.
Cancer cachexia has long been difficult to treat. Nutritional support alone is often ineffective because the condition is driven by metabolic and inflammatory changes rather than a lack of calories.
Current treatments are limited, and no drugs are approved in the United States specifically to prevent cancer-related muscle wasting.
“MESH1 controls a newly discovered protective function of coenzyme A that helps determine how muscle cells respond to ferroptosis,” said one of first authors Chao-Chieh Lin, PhD, of the Duke Department of Molecular Genetics and Microbiology. “This opens the door to new treatments for cancer cachexia.”
First authors of the study include Chao-Chich Lin, Joshua Rose, and Alexander A Mestre.
Researchers received support from the National Institutes of Health, Duke DCI Pilot Project, Department of Defense, Singapore’s Ministry of Education AcRF Award, Diana Koh Innovative Cancer Research Award, the National Medical Research Council and Singapore Ministry of Health.